|
Therefore, it is the interplay between all of the factors affecting these flows (i.e., microvascular pressure, lymphatic contractility, serosal permeability, etc.) that determines the steady-state interstitial fluid pressure ( 6). An increase in interstitial fluid pressure leads to a decrease in microvascular filtration and to increases in lymph flow and trans-serosal flow. All of these fluid flows–microvascular filtration, lymph flow, and trans-serosal flow–are significantly influenced by interstitial fluid (hydrostatic) pressure. In the edematous intestine, interstitial fluid can cross the mucosal barrier into the lumen ( 4, 5). 
In organs located in the pleural, pericardial and peritoneal spaces, some interstitial fluid filters through the organ's serosal surface into the surrounding fluid space and, then, is also taken up into the lymphatic system. Interstitial fluid is removed via lymphatic drainage and returned to the venous circulation. Notable exceptions include the peritubular capillaries in the kidney and microvascular beds within intestinal villi that routinely absorb interstitial fluid ( 2, 3). 
Under most conditions in most tissues, fluid from the vascular space continually filters from the microvessels into the interstitial space and is not reabsorbed ( 1). The interstitial space that lies between blood vessels and cells provides the fluid and structural environment surrounding those cells. This review examines current concepts regarding regulation of interstitial volume, pressure and flow and, utilizing that background, addresses topics of interest that impact IV fluid administration. A third avenue of inquiry has confirmed that the intestinal interstitial space and mesenteric lymphatic system provide a critical link between circulatory shock and the subsequent systemic inflammatory response syndrome. On another front, recent investigations have revealed that the interstitial space plays an active role, in addition to the kidney, in the regulation of salt and water balance in the body including blood pressure. We now know, however, that the generally predictable relationship between interstitial volume and interstitial pressure can change markedly and that these transitional episodes can occur within minutes. The passive view holds that changes in interstitial volume and pressure are the result of influences outside of the interstitium, i.e., microvascular pressure, microvascular permeability or plasma colloid osmotic pressure. 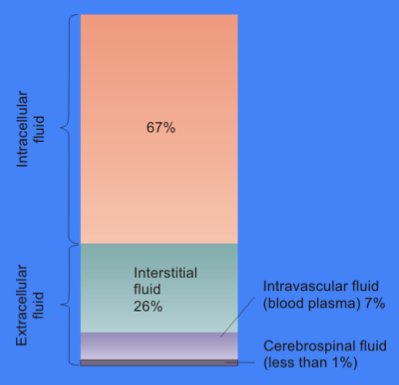
The traditional view that the interstitial space is a relatively static and, frankly, uninteresting region is being reassessed in light of new research showing that the interstitium plays an active role in the regulation of interstitial volume and content and is a key participant in the pathogenesis of inflammation and shock. Lastly, current evidence strongly supports the non-intuitive view that the primary factor leading to inflammatory edema formation is a decrease in interstitial hydrostatic pressure that dramatically increases microvascular filtration. 
Second, it now appears that hypovolemic shock leads to systemic inflammatory response syndrome principally through the entry of digestive enzymes into the intestinal interstitial space and the subsequent progression of enzymes and inflammatory agents through the mesenteric lymphatic system to the general circulation. This led to the hypothesis that, along with the kidney, the interstitial space plays an active role in the long-term regulation of blood pressure. The first of these started with the discovery that excess dietary salt can be stored non-osmotically in the interstitial space with minimal impact on vascular volume and pressures. This review examines current concepts regarding regulation of interstitial volume, pressure, and flow and utilizes that background to address three major topics of interest that impact IV fluid administration. These difficulties have long obscured what we are beginning to appreciate is a dynamic milieu that is subject to both intrinsic and extrinsic regulation. However, the contents and the hydrostatic pressure of this interstitial fluid can be very difficult to determine even in experimental settings. Increases in the volume of the interstitial space are readily recognized clinically as interstitial edema formation in the loose connective tissue of skin, mucosa, and lung.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
